Efficacy and Safety of Sonic Hedgehog Inhibitors and PD-1 Inhibitors in Locally Advanced Basal Cell Carcinoma Management: A Systematic Review and Meta-analysis (2013-2023)
DOI:
https://doi.org/10.59796/jcst.V15N2.2025.110Keywords:
basal cell carcinoma, systematic review, meta-analysis, sonic Hedgehog inhibitors, PD-1 inhibitorsAbstract
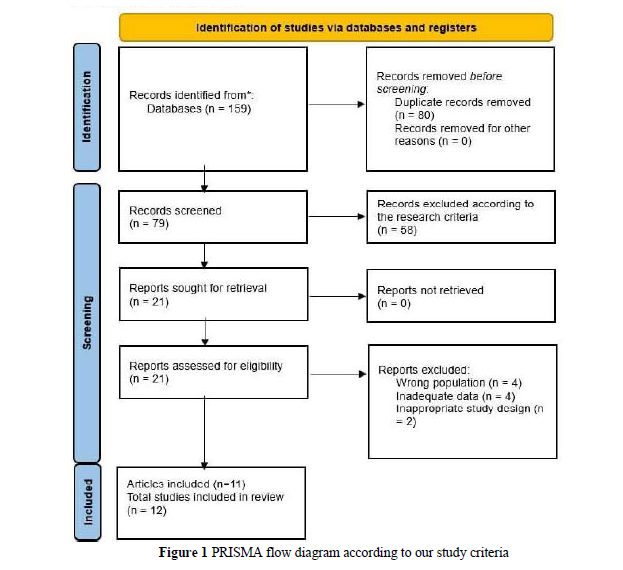
The management of advanced basal cell carcinoma (aBCC), in contrast to non-advanced BCC, is often a significant challenge for patients and treating physicians. Nevertheless, sonic Hedgehog inhibitors and, more recently, immune checkpoint inhibitors have offered new hope for improved clinical outcomes. A thorough evaluation of the potential adverse effects of these systemic therapies is also crucial. This review provides detailed information on the clinical efficacy and safety of various regimens of sonic Hedgehog pathway inhibitors and immune checkpoint inhibitors in locally advanced basal cell carcinoma (laBCC) management over the last decade. Our systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. We analyzed the data specific to patients with laBCC who received Hedgehog pathway and immune checkpoint inhibitors between 2013 and 2023 and presented the outcomes accordingly. Eleven articles were included in our systematic review, and ten articles were eligible for overall response rate (ORR) and complete response rate (CRR) meta-analysis. ORRs for vismodegib, sonidegib, cemiplimab, and nivolumab were 74%, 50%, 31%, and 17%, respectively. The complete response rate (CRR) was significantly higher for vismodegib at 40%, compared to sonidegib (2%) and cemiplimab (6%). The most common adverse effects of hedgehog pathway inhibitors include muscle spasms, dysgeusia, and alopecia, while cemiplimab is frequently associated with fatigue, diarrhea, and pruritus. The systemic therapies present a promising approach for the management of laBCC; however, their use is often limited by adverse effects. Among available options, vismodegib demonstrates superior ORR and CRR compared to sonidegib and immunotherapy, highlighting its potential as a preferred option.
References
Basset-Séguin, N., Hauschild, A., Kunstfeld, R., Grob, J., Dréno, B., Mortier, L., ... & Hansson, J. (2017). Vismodegib in patients with advanced basal cell carcinoma: Primary analysis of STEVIE, an international, open-label trial. European Journal of Cancer, 86, 334–348. https://doi.org/10.1016/j.ejca.2017.08.022
Bertrand, N., Guerreschi, P., Basset-Seguin, N., Saiag, P., Dupuy, A., Dalac-Rat, S., ... & Mortier, L. (2021). Vismodegib in neoadjuvant treatment of locally advanced basal cell carcinoma: First results of a multicenter, open-label, phase 2 trial (VISMONEO study): Neoadjuvant Vismodegib in Locally Advanced Basal Cell Carcinoma. EClinicalMedicine, 35, Article 100844. https://doi.org/10.1016/j.eclinm.2021.100844
Bichakjian, C., Armstrong, A., Baum, C., Bordeaux, J. S., Brown, M., Busam, K. J., ... & Rodgers, P. (2018). Guidelines of care for the management of basal cell carcinoma. Journal of the American Academy of Dermatology, 78(3), 540-559. https://doi.org/10.1016/j.jaad.2017.10.006
Danial, C., Sarin, K. Y., Oro, A. E., & Chang, A. L. S. (2016). An Investigator-Initiated Open-Label Trial of Sonidegib in Advanced Basal Cell Carcinoma Patients Resistant to Vismodegib. Clinical Cancer Research: An Official Journal of the American Association for Cancer Research, 22(6), 1325–1329. https://doi.org/10.1158/1078-0432.CCR-15-1588
De Giorgi, V., Scarfì, F., Trane, L., Silvestri, F., Venturi, F., Zuccaro, B., ... & Longo, C. (2021). Treatment of Advanced Basal Cell Carcinoma with Hedgehog Pathway Inhibitors: A Multidisciplinary Expert Meeting. Cancers, 13(22), Article 5706. https://doi.org/10.3390/cancers13225706
Dummer, R., Guminksi, A., Gutzmer, R., Lear, J. T., Lewis, K. D., Chang, A. L. S., ... & Migden, M. R. (2020). Long-term efficacy and safety of sonidegib in patients with advanced basal cell carcinoma: 42-month analysis of the phase II randomized, double-blind BOLT study. The British Journal of Dermatology, 182(6), 1369–1378. https://doi.org/10.1111/bjd.18552
Fecher, L. A. (2013). Systemic therapy for inoperable and metastatic basal cell cancer. Current Treatment Options in Oncology, 14(2), 237–248. https://doi.org/10.1007/s11864-013-0233-9
Kahana, A., Unsworth, S. P., Andrews, C. A., Chan, M. P., Bresler, S. C., Bichakjian, C. K., ... & Worden, F. P. (2021). Vismodegib for Preservation of Visual Function in Patients with Advanced Periocular Basal Cell Carcinoma: The VISORB Trial. The Oncologist, 26(7), e1240–e1249. https://doi.org/10.1002/onco.13820
Ketkomol, P., Songsak, T., Jongrungruangchok, S., Madaka, F., & Pradubyat, N. (2024). The effect of 1'-acetoxychavicol acetate on A549 human non-small cell lung cancer. Journal of Current Science and Technology,14(2), Article 43. https://doi.org/10.59796/jcst.V14N2.2024.43
Lear, J. T., Corner, C., Dziewulski, P., Fife, K., Ross, G. L., Varma, S., & Harwood, C. A. (2014). Challenges and new horizons in the management of advanced basal cell carcinoma: A UK perspective. British Journal of Cancer, 111(8), 1476–1481. https://doi.org/10.1038/bjc.2014.270
Niebel, D., Sirokay, J., Hoffmann, F., Fröhlich, A., Bieber, T., & Landsberg, J. (2020). Clinical Management of Locally Advanced Basal-Cell Carcinomas and Future Therapeutic Directions. Dermatology and Therapy, 10(4), 835–846. https://doi.org/10.1007/s13555-020-00382-y
Oh, C. C., Jin, A., & Koh, W.-P. (2021). Trends of cutaneous basal cell carcinoma, squamous cell carcinoma, and melanoma among the Chinese, Malays, and Indians in Singapore from 1968-2016. JAAD International, 4, 39–45. https://doi.org/10.1016/j.jdin.2021.05.006
Rojanamatin, J., Ukranun, W., Supaattagorn, P., Chiawiriyabunya, I., Wongsena, M., Chaiwerawattana, A., … & Buasom, R. (Eds.). (2021). Cancer in Thailand: Vol. X, 2016–2018. Medical Record and Databased Cancer Unit.
Saelee, P., Pongtheerat, T., Sophonnithiprasert, T., & Jinda, W. (2022). Clinicopathological significance of FANCAmRNA expression in Thai patients with breast cancer. Journal of Current Science and Technology, 12(3), 408-416. https://doi.org/10.14456/jcst.2022.31
Sekulic, A., Migden, M. R., Basset-Seguin, N., Garbe, C., Gesierich, A., Lao, C. D., ... & ERIVANCE BCC Investigators. (2017). Long-term safety and efficacy of vismodegib in patients with advanced basal cell carcinoma: Final update of the pivotal ERIVANCE BCC study. BMC Cancer, 17(1), Article 332. https://doi.org/10.1186/s12885-017-3286-5
Sekulic, A., Yoo, S., Kudchadkar, R., Guillen, J., Rogers, G., Chang, A. L. S., ... & Lacouture, M. (2022). Real-world assessment and treatment of locally advanced basal cell carcinoma: Findings from the RegiSONIC disease registry. PLOS ONE, 17(1), Article e0262151. https://doi.org/10.1371/journal.pone.0262151
Skin Cancer Facts & Statistics. (n.d.). The Skin Cancer Foundation. Retrieved June 3, 2023, from https://www.skincancer.org/skin-cancer-information/skin-cancer-facts/
Stratigos, A. J., Sekulic, A., Peris, K., Bechter, O., Prey, S., Kaatz, M., ... & Fury, M. G. (2021). Cemiplimab in locally advanced basal cell carcinoma after hedgehog inhibitor therapy: an open-label, multi-centre, single-arm, phase 2 trial. The Lancet Oncology, 22(6), 848-857. https://doi.org/10.1016/S1470-2045(21)00126-1
Verkouteren, J. A. C., Ramdas, K. H. R., Wakkee, M., & Nijsten, T. (2017). Epidemiology of basal cell carcinoma: scholarly review. British Journal of Dermatology, 177(2), 359-372. https://doi.org/10.1111/bjd.15321
Véron, M., Chevret, S., Grob, J. J., Beylot-Barry, M., Saiag, P., Fléchon, A., ... & Mortier, L. (2022). Safety and efficacy of nivolumab, an anti-PD1 immunotherapy, in patients with advanced basal cell carcinoma, after failure or intolerance to sonic Hedgehog inhibitors: UNICANCER AcSé NIVOLUMAB trial. European Journal of Cancer, 177, 103-111. https://doi.org/10.1016/j.ejca.2022.09.013
Weinstock, M. A., & Still, J. M. (2011). Assessing current treatment options for patients with severe/advanced basal cell carcinoma. Seminars in Cutaneous Medicine and Surgery, 30(4 Suppl), S10-13. https://doi.org/10.1016/j.sder.2011.11.004
Xavier, C., Lopes, E., Bexiga, C., Moura, C., Gouveia, E., & Duarte, A. F. (2021). Vismodegib for treatment of periocular basal cell carcinoma – 6-year experience from a tertiary cancer center. Anais Brasileiros de Dermatologia, 96(6), 712–716. https://doi.org/10.1016/j.abd.2021.04.012
Yenchitsomanus, P. (2024). Cancer Immunotherapy: Challenges and Advancements in CAR T Cell Technology. Siriraj Medical Journal, 76(5), 313–324. https://doi.org/10.33192/smj.v76i5.268031
Downloads
Published
How to Cite
License
Copyright (c) 2025 Journal of Current Science and Technology

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.