AVCSNPs, a novel alternative antibiotic derived from chitosan nanoparticles loaded with Aloe Vera flavonoids
DOI:
https://doi.org/10.59796/jcst.V16N2.2026.174Keywords:
chitosan nanoparticles, Aloe vera flavonoids, aac(6′)-Ie-aph(2”)-Ia, mecA gene, green synthesis, AVCSNPsAbstract
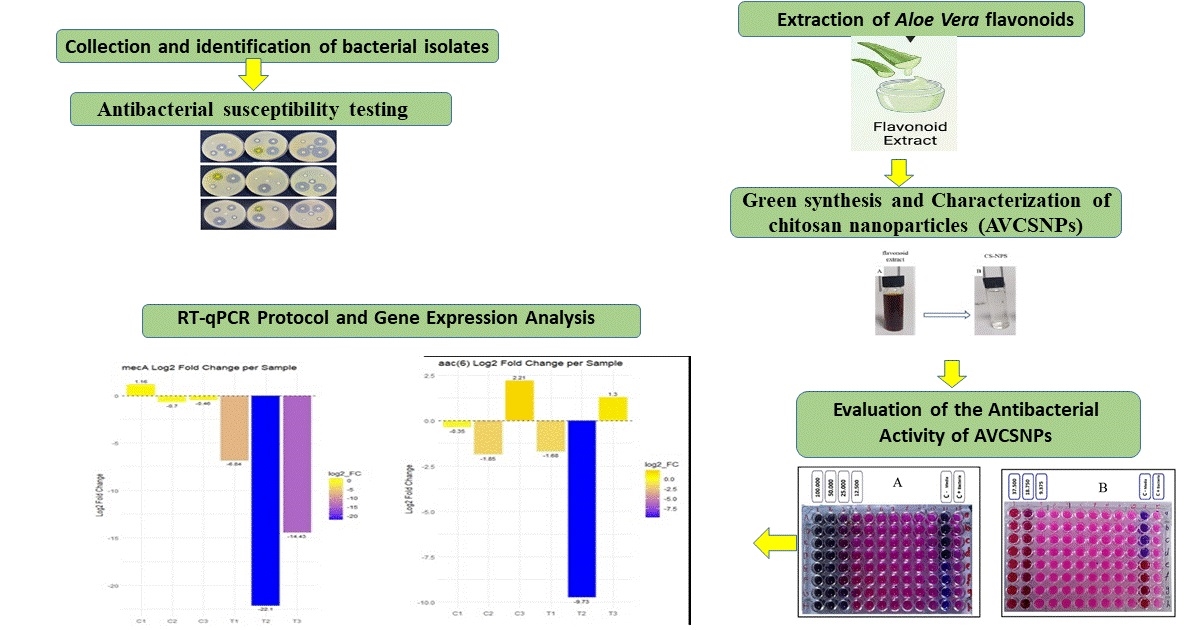
The treatment of burn and wound infections is becoming more challenging due to the emergence of antibiotic-resistant bacteria. This study investigated the synergistic antibacterial efficacy of green-synthesized chitosan nanoparticles (CSNPs) loaded with Aloe vera gel flavonoid extract (designated AVCSNPs), which were effective against MDR and XDR Staphylococcus aureus isolates. The primary goal was to evaluate the antibacterial efficacy of AVCSNPs compared with flavonoid extract alone. Using AVCSNPs as an alternative to antibiotics for Staphylococcus aureus is a low-toxicity and cost-effective approach. The study also examined how AVCSNPs affected the expression of genes associated with antibiotic resistance, such as mecA and aac(6′)-Ie-aph(2″)-Ia. Clinical samples were obtained from Ghazi Al-Hariri Hospital and Burns Hospital in Baghdad. The MIC of the flavonoid extract was determined. AVCSNPs were characterized by UV-Vis spectroscopy and particle size analysis after biosynthesis. Using log2-fold change analysis, the effects of treatment on gene expression were investigated. AVCSNPs exhibited greater antibacterial activity than the flavonoid extract, with a MIC of 18.75 µg/mL compared with 50 µg/mL. Studying genes affected by AVCSNPs was essential for understanding antibiotic resistance. Treatment with AVCSNPs significantly reduced the expression of the mecA gene, with a mean log2-fold decrease of -14.64. This notable decline indicates that nanoparticles may circumvent the primary resistance mechanism in MRSA bacteria. However, although the decline was less pronounced (-3.37), the expression of the aac(6′)-Ie-aph(2″)-Ia gene also declined. Due to their potent and targeted action on the mecA gene, AVCSNPs may be a viable and biocompatible alternative to conventional antibiotics for the treatment of MRSA infections.
References
Akdaşçi, E., Duman, H., Eker, F., Bechelany, M., & Karav, S. (2025). Chitosan and its nanoparticles: A multifaceted approach to antibacterial applications. Nanomaterials, 15(2), Article 126. https://doi.org/10.3390/nano15020126
Ali, A. H. (2022). High-performance liquid chromatography (HPLC): A review. Annals of Advances in Chemistry, 6(1), 10-20. https://doi.org/10.29328/journal.aac.1001026
Ali, M. R., & Khudhair, A. M. (2018). Detection of colony adhesion factors and genetic background of adhesion genes among multidrug-resistant uropathogenic Escherichia coli Isolated in Iraq. Journal of Pure and Applied Microbiology, 12(4), 2017-2025. https://doi.org/10.22207/JPAM.12.4.38
Al-Khafaji, M. M. S., & Al-Hayawi, A. Y. (2024). Deciphering the impact of miR-92a-3p on controlling BCL11A gene expression and its implication in easing thalassemia symptoms. Medical Journal of Babylon, 21(Suppl 2), S250-S257. https://doi.org/10.4103/MJBL.MJBL_1337_23
Al-Kubaisi, Z. A., Al-Shmgani, H. S., & Salman, M. J. (2020). Evaluation of in vivo and in vitro protective effects of quercetin on lipopolysaccharide-induced inflammation and cytotoxicology. Research Journal of Pharmacy and Technology, 13(8), 3897-3902. http://doi.org/10.5958/0974-360X.2020.00690.3
AL-Lami, R. A., Al-Hayanni, H. S., & Shehab, Z. H. (2022). Molecular investigation of some beta-lactamase genes by PCR and DNA sequencing techniques in clinical Escherichia coli. Iraqi Journal of Science, 63(10), 4205-4212. https://doi.org/10.24996/ijs.2022.63.10.7
Al-Musawi, M. M., Al-Bairuty, G. A., & Al-Shmgani, H. S. (2022). The comparative effect of copper oxide nanoparticles and copper sulfate on reproductive hormones and sperm parameters in mature male albino mice. Annals of Biology, 38(2), 317-321. https://doi.org/10.1155/2022/4877637
Aldhahir, S., Al-Rayahi, I. A. M., & Muhsin, S. S. (2025). Comparison of myxovirus resistance gene 2 expression among adult and juvenile SLE Iraqi patients. Journal of Current Science and Technology, 15(2), Article 97. https://doi.org/10.59796/jcst.V15N2.2025.97
Amini, S., Omidi, A. H., Afkhami, H., Sabati, H., Mohsenzadeh, A., Soleymani, A., ... & Mohammadi, M. R. (2024). Frequency of aac(6 ́)-le-aph (2 ́ ́) gene and resistance to Aminoglycoside antibiotics in Staphylococcus aureus isolates. Cellular, Molecular and Biomedical Reports, 4(2), 120-128. https://doi.org/10.55705/cmbr.2023.411361.1166
Ausaj, A. A., Al-Shmgani, H. S., Abid, W. B., Gadallah, A. A., Mashlawi, A. M., Khormi, M. A., ... & Abada, E. (2024). Immobilization of bromelain on gold nanoparticles for comprehensive detection of their antioxidant, anti-angiogenic, and wound-healing potentials. Inorganics, 12(12), Article 325. https://doi.org/10.3390/inorganics12120325
Bouslamti, M., El Barnossi, A., Kara, M., Alotaibi, B. S., Al Kamaly, O., Assouguem, A., ... & Benjelloun, A. S. (2022). Total polyphenols content, antioxidant and antimicrobial activities of leaves of Solanum elaeagnifolium Cav. from Morocco. Molecules, 27(13), Article 4322. https://doi.org/10.3390/molecules27134322
Choi, S. M., Kim, S. H., Kim, H. J., Lee, D. G., Choi, J. H., Yoo, J. H., ... & Kang, M. W. (2003). Multiplex PCR for the detection of genes encoding aminoglycoside modifying enzymes and methicillin resistance among Staphylococcus species. Journal of Korean Medical Science, 18(5), Article 631. https://doi.org/10.3346/jkms.2003.18.5.631
Cushnie, T. T., & Lamb, A. J. (2011). Recent advances in understanding the antibacterial properties of flavonoids. International Journal of Antimicrobial Agents, 38(2), 99-107. https://doi.org/10.1016/j.ijantimicag.2011.02.014
Eckmann, C., & Dryden, M. (2010). Treatment of complicated skin and soft-tissue infections caused by resistant bacteria: Value of linezolid, tigecycline, daptomycin and vancomycin. European Journal of Medical Research, 15(12), Article 554. https://doi.org/10.1186/2047-783X-15-12-554
El-Naggar, N. E. A., Shiha, A. M., Mahrous, H., & Mohammed, A. A. (2022). Green synthesis of chitosan nanoparticles, optimization, characterization and antibacterial efficacy against multi drug resistant biofilm-forming Acinetobacter baumannii. Scientific Reports, 12(1), Article 19869. https://doi.org/10.1038/s41598-022-24303-5
Ender, M., McCallum, N., & Berger-Bächi, B. (2008). Impact of mecA promoter mutations on mecA expression and β-lactam resistance levels. International Journal of Medical Microbiology, 298(7-8), 607-617. https://doi.org/10.1016/j.ijmm.2008.01.015
Faiq, N. H., & Ahmed, M. E. (2024). Inhibitory effects of biosynthesized copper nanoparticles on biofilm formation of Proteus mirabilis. Iraqi Journal of Science, 65(1), 65-78. https://doi.org/10.24996/ijs.2024.64.1.7
Fishovitz, J., Hermoso, J. A., Chang, M., & Mobashery, S. (2014). Penicillin‐binding protein 2a of methicillin‐resistant Staphylococcus aureus. International Union of Biochemistry and Molecular Biology life, 66(8), 572-577. https://doi.org/10.1002/iub.1289
Gidamo, G. H. (2024). Stem cells of Impatiens tinctoria A. Rich tuber and antioxidant activity of their extracts. Journal of Current Science and Technology, 14(3), Article 59. https://doi.org/10.59796/jcst.V14N3.2024.59
Hauschild, T., Sacha, P., Wieczorek, P., Zalewska, M., Kaczyńska, K., & Tryniszewska, E. (2008). Aminoglycosides resistance in clinical isolates of Staphylococcus aureus from a University Hospital in Bialystok, Poland. Folia Histochemica et Cytobiologica, 46(2), 225-228. https://doi.org/10.2478/v10042-008-0034-3
Huanbutta, K., Sriamornsak, P., Suwanpitak, K., Klinchuen, N., Deebugkum, T., Teppitak, V., & Sangnim, T. (2023). Key fabrications of chitosan nanoparticles for effective drug delivery using flow chemistry reactors. International Journal of Nanomedicine, 18, 7889–7900. https://doi.org/10.2147/IJN.S433756
Humphries, R., Bobenchik, A. M., Hindler, J. A., & Schuetz, A. N. (2021). Overview of changes to the Clinical and Laboratory Standards Institute performance standards for antimicrobial susceptibility testing, M100, 31st edition. Journal of Clinical Microbiology, 59(10), Article e00213-21. https://doi.org/10.1128/jcm.00213-21
Hussain, A. I., Anwar, F., Nigam, P. S., Sarker, S. D., Moore, J. E., Rao, J. R., & Mazumdar, A. (2011). Antibacterial activity of some Lamiaceae essential oils using resazurin as an indicator of cell growth. LWT-Food Science and Technology, 44(4), 1199-1206. https://doi.org/10.1016/j.lwt.2010.10.005
Jafar, F. N., & Sabah, F. S. (2024). Isolation of a flavonoid bioactive compound from Gymnoascus dankaliensis Secondary Metabolite. Iraqi Journal of Science, 65(12), 6932-6945. https://doi.org/10.24996/ijs.2024.65.12.11
Jirakitticharoen, S., Wisuitiprot, W., Jitareerat, P., & Wongs-Aree, C. (2022). Phenolics, antioxidant and antibacterial activities of immature and mature Blumea balsamifera leaf extracts eluted with different solvents. Journal of Tropical Medicine, 2022(1), Article 7794227. https://doi.org/10.1155/2022/7794227
Kadium, S. W., & Sahib, R. A. (2023). Antifungal activity of phenols compound separated from quercus infectoria and citrullus colocynthis against toxic fungi. Archives of Razi Institute, 78(1), Article 297. https://doi.org/10.22092/ARI.2022.358960.2347
Karuncharoenpanich, T., Phetmanee, T., Pradubyat, N., Songsak, T., Jongrungruangchok, S., Madaka, F., Wunnakup, T., & Lakkana, N. (2025). In vitro assessment of antimicrobial activity and synergistic effects of ethanolic extracts from six medicinal plants. Journal of Current Science and Technology, 15(1), Article 85. https://doi.org/10.59796/jcst.V15N1.2025.85
Kiani, D. (2023). X-ray diffraction (XRD). Springer Handbook of Advanced Catalyst Characterization. Cham, Switzerland: Springer International Publishing. https://doi.org/10.1007/978-3-031-07125-6_25
Mehrotra, M., Wang, G., & Johnson, W. M. (2000). Multiplex PCR for detection of genes for Staphylococcus aureus enterotoxins, exfoliative toxins, toxic shock syndrome toxin 1, and methicillin resistance. Journal of Clinical Microbiology, 38(3), 1032-1035. https://doi.org/10.1128/jcm.38.3.1032-1035.2000
Mendem, S. K., Alasthimannahalli Gangadhara, T., Shivannavar, C. T., & Gaddad, S. M. (2016). Antibiotic resistance patterns of Staphylococcus aureus: A multi-centre study from India. Microbial Pathogenesis, 98, 167–170. https://doi.org/10.1016/j.micpath.2016.07.010
Mishra, B. B. T. S., Patel, B. B., & Tiwari, S. (2010). Colloidal nanocarriers: A review on formulation technology, types and applications toward targeted drug delivery. Nanomedicine: Nanotechnology, Biology and Medicine, 6(1), 9-24. https://doi.org/10.1016/j.nano.2009.04.008
Morovat, T., Bahram, F., Mohammad, E., Setareh, S., & Mohamad Mehdi, F. (2009). Distribution of different carbapenem resistant clones of Acinetobacter baumannii in Tehran hospitals. The New Microbiologica, 32(3), 265-271.
O'Brien, J., Wilson, I., Orton, T., & Pognan, F. (2000). Investigation of the Alamar Blue (resazurin) fluorescent dye for the assessment of mammalian cell cytotoxicity. European Journal of Biochemistry, 267(17), 5421–5426. https://doi.org/10.1046/j.1432-1327.2000.01606.x
Prisa, D. (2022). Aloe: Medicinal properties and botanical characteristics. Journal of Current Science and Technology, 12(3), 605–614. https://doi.org/10.14456/jcst.2022.46
Rahman, A., Kafi, M. A., Beak, G., Saha, S. K., Roy, K. J., Habib, A., ... & Choi, J. W. (2024). Green-synthesized chitosan nanoparticles for controlling multidrug-resistant mecA-and blaZ-positive Staphylococcus aureus and aadA1-positive Escherichia coli. International Journal of Molecular Sciences, 25(9), Article 4746. https://doi.org/10.3390/ijms25094746
Urtgam, S., Thawathotsapakorn, S., & Wattanachaiyingcharoen, W. (2024). The partial nucleotide sequences of the mitochondrial genes, COI and 16S rRNA, of fireflies in the genera Pygoluciola, Trisinuata, and Medeopteryx (Coleoptera: Lampyridae). Journal of Current Science and Technology, 14(2), Article 22. https://doi.org/10.59796/jcst.V14N2.2024.22
Vercelli, C., Amadori, M., Gambino, G., & Re, G. (2023). Does nitrofurantoin improve the portfolio of vets against resistant bacteria in companion animals?. Antibiotics, 12(5), Article 911. https://doi.org/10.3390/antibiotics12050911
Villarta, J. D. A., Paylago, F. J. C., Poldo, J. C. H., Santos, J. S. R., Escordial, T. A. M. M., & Montealegre, C. M. (2025). Green synthesis, characterization, and optimization of chitosan nanoparticles using blumea balsamifera extract. Processes, 13(3), Article 804. https://doi.org/10.3390/pr13030804
World Health Organization. (2022). Global antimicrobial resistance and use surveillance system (GLASS) report 2022. Retrieved from https://apps.who.int/iris/handle/10665/364996
Zhang, D., Cui, K., Wang, T., Dong, H., Feng, W., Ma, C., & Dong, Y. (2018). Trends in and correlations between antibiotic consumption and resistance of Staphylococcus aureus at a tertiary hospital in China before and after introduction of an antimicrobial stewardship programme. Epidemiology and Infection, 147, Article e48. https://doi.org/10.1017/S0950268818003059
Downloads
Published
How to Cite
License
Copyright (c) 2026 Journal of Current Science and Technology

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.