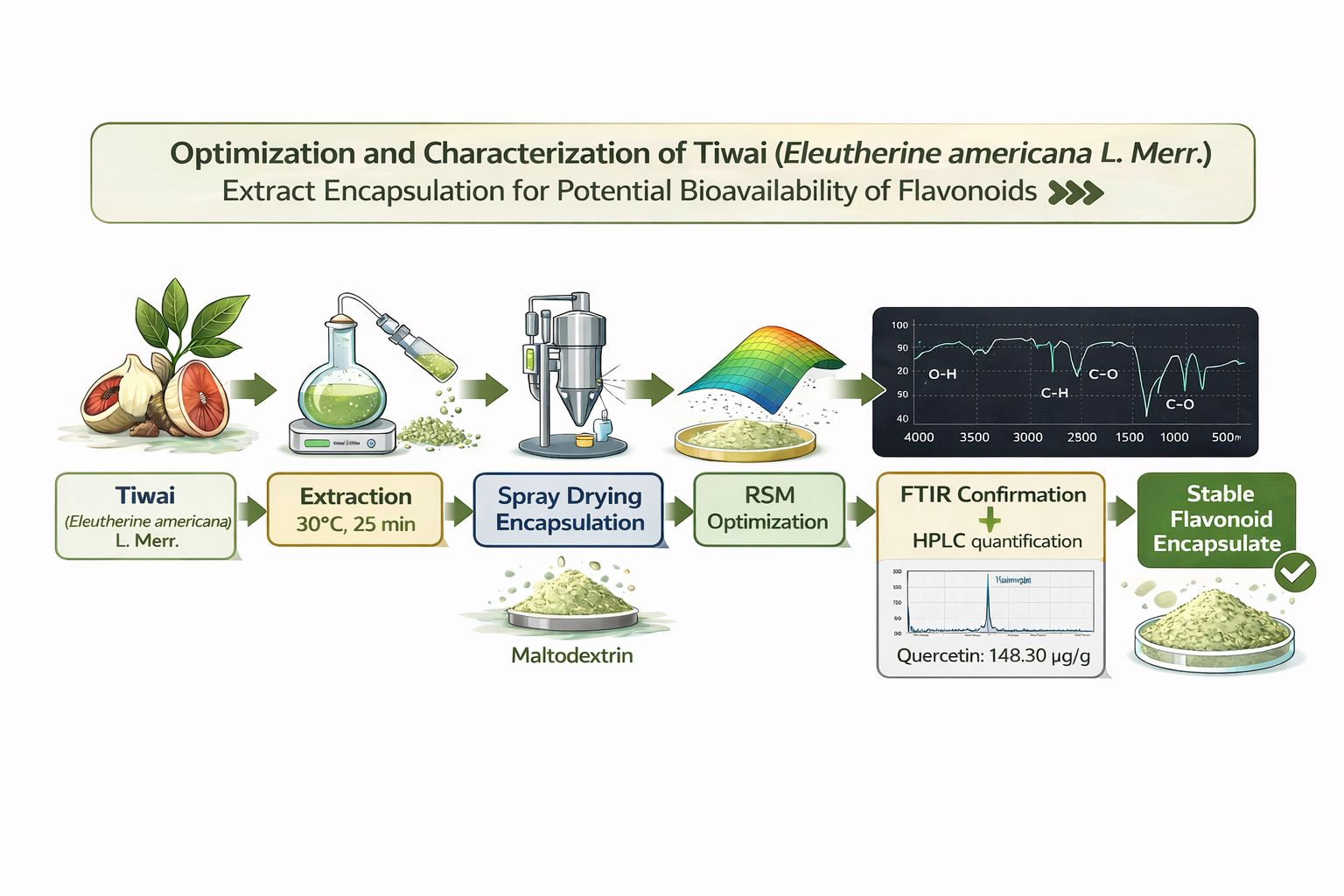
Optimization and Characterization of Tiwai (Eleutherine americana L. Merr.) Extract Encapsulation for Potential Bioavailability of Flavonoids
DOI:
https://doi.org/10.59796/jcst.V16N2.2026.172Keywords:
antioxidant, encapsulation, flavonoids, spray drying, FTIR, HPLCAbstract
Tiwai (Eleutherine americana L. Merr.) is a traditional medicinal plant from Kalimantan, rich in bioactive compounds, particularly flavonoids, which exhibit strong antioxidant activity and therapeutic benefits. However, flavonoids in free form tend to be unstable, making encapsulation necessary to enhance their stability, bioavailability, and shelf life. This study optimized the encapsulation of Tiwai extract using the spray drying method, with parameters such as the ratio of Tiwai extract to maltodextrin (ET:MD), chitosan concentration, and drying inlet temperature adjusted using Response Surface Methodology (RSM) with Box-Behnken Design (BBD). The study provided detailed results, including an encapsulation efficiency (EE) of 93.24%, loading capacity (LC) of 95.76%, total flavonoid content (TFC) of 69.23 mg QE/g, and antioxidant activity (DPPH) of 60.01 µg/mL. A significant discrepancy was found between the quercetin content determined by HPLC (148.30 µg/g) and the TFC obtained using the AlCl₃ method (69.23 mg QE/g). This difference is attributed to the different principles of the two methods, as the AlCl₃ method reacts with flavonoids and other antioxidant compounds, whereas HPLC specifically identifies quercetin. FTIR analysis confirmed successful encapsulation, showing changes in the functional groups of the encapsulated product. These findings suggest that the optimized Tiwai encapsulation formula has potential applications in functional foods and pharmaceuticals.
References
Adesina, S., Iwalewa, E., & Johnny, I. (2016). Tetrapleura tetraptera Taub-ethnopharmacology, chemistry, medicinal and nutritional values-a review. British Journal of Pharmaceutical Research, 12(3), 1-22. https://doi.org/10.9734/BJPR/2016/26554
Akbarbaglu, Z., Jafari, S. M., Sarabandi, K., Mohammadi, M., Heshmati, M. K., & Pezeshki, A. (2019). Influence of spray drying encapsulation on the retention of antioxidant properties and microstructure of flaxseed protein hydrolysates. Colloids and Surfaces B: Biointerfaces, 178, 421-429. https://doi.org/10.1016/j.colsurfb.2019.03.038
Castro-López, C., Espinoza-González, C., Ramos-González, R., Boone-Villa, V. D., Aguilar-González, M. A., Martínez-Ávila, G. C., ... & Ventura-Sobrevilla, J. M. (2021). Spray-drying encapsulation of microwave-assisted extracted polyphenols from Moringa oleifera: Influence of tragacanth, locust bean, and carboxymethyl-cellulose formulations. Food Research International, 144, Article 110291. https://doi.org/10.1016/j.foodres.2021.110291
Chen, P., Cui, H., Feng, L., Yu, J., Hayat, K., Jia, C., ... & Ho, C. T. (2022). Effect of the C-ring structure of flavonoids on the yield of adducts formed by the linkage of the active site at the A-ring and Amadori rearrangement products during the Maillard intermediate preparation. Journal of Agricultural and Food Chemistry, 70(10), 3280-3288. https://doi.org/10.1021/acs.jafc.1c07521
Chen, Y., Mastalerz, M., & Schimmelmann, A. (2012). Characterization of chemical functional groups in macerals across different coal ranks via micro-FTIR spectroscopy. International Journal of Coal Geology, 104, 22-33. https://doi.org/10.1016/j.coal.2012.09.001
Chew, Y. L., Goh, J. K., & Lim, Y. Y. (2009). Assessment of in vitro antioxidant capacity and polyphenolic composition of selected medicinal herbs from Leguminosae family in Peninsular Malaysia. Food Chemistry, 116(1), 13-18. https://doi.org/10.1016/j.foodchem.2009.01.091
Cocuron, J. C., Casas, M. I., Yang, F., Grotewold, E., & Alonso, A. P. (2019). Beyond the wall: High-throughput quantification of plant soluble and cell-wall bound phenolics by liquid chromatography tandem mass spectrometry. Journal of Chromatography A, 1589, 93-104. https://doi.org/10.1016/j.chroma.2018.12.059
Deshmukh, M. D., & Patil, M. P. (2024). The quality by design approach for analytical method development and validation of the RP-HPLC method for estimation of quercetin in pure, marketed, and cream formulation. Biosciences Biotechnology Research Asia, 21(1), Article 273. https://doi.org/10.13005/bbra/3224
Ding, J., Xu, Z., Qi, B., Cui, S., Wang, T., Jiang, L., ... & Sui, X. (2019). Fabrication and characterization of soybean oil bodies encapsulated in maltodextrin and chitosan-EGCG conjugates: An in vitro digestibility study. Food Hydrocolloids, 94, 519-527. https://doi.org/10.1016/j.foodhyd.2019.04.001
Fornaro, T., Biczysko, M., Bloino, J., & Barone, V. (2016). Reliable vibrational wavenumbers for C [double bond, length as m-dash] O and N–H stretchings of isolated and hydrogen-bonded nucleic acid bases. Physical Chemistry Chemical Physics, 18(12), 8479-8490. https://doi.org/10.1039/c5cp07386c
Gulcin, İ. (2020). Antioxidants and antioxidant methods: An updated overview. Archives of Toxicology, 94(3), 651-715. https://doi.org/10.1007/s00204-020-02689-3
Hao, J., Fang, X., Zhou, Y., Wang, J., Guo, F., Li, F., & Peng, X. (2011). Development and optimization of solid lipid nanoparticle formulation for ophthalmic delivery of chloramphenicol using a Box-Behnken design. International Journal of Nanomedicine, 6, 683-692. https://doi.org/10.2147/IJN.S17386
Hong, S., Shen, S., Tan, D. C. T., Ng, W. K., Liu, X., Chia, L. S., ... & Gokhale, R. (2016). High drug load, stable, manufacturable and bioavailable fenofibrate formulations in mesoporous silica: A comparison of spray drying versus solvent impregnation methods. Drug Delivery, 23(1), 316-327. https://doi.org/10.3109/10717544.2014.913323
Hongsing, P., Kongart, C., Nuiden, N., Wannigama, D. L., & Phairoh, K. (2024). Quantitative analysis of swertiamarin content from Fagraea fragrans leaf extract using HPLC technique and its correlation to antibacterial activity. Journal of Current Science and Technology, 14(2), Article 34. https://doi.org/10.59796/jcst.V14N2.2024.341
Horincar, G., Aprodu, I., Barbu, V., Râpeanu, G., Bahrim, G. E., & Stănciuc, N. (2019). Interactions of flavonoids from yellow onion skins with whey proteins: Mechanisms of binding and microencapsulation with different combinations of polymers. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 215, 158-167. https://doi.org/10.1016/j.saa.2019.02.100
Jain, V. A. N. D. A. N. A., & Shaikh, M. S. (2016). Simultaneous RP-HPLC analysis of quercetin and kaempferol in different plant parts of cissus quadrangularis. International Journal of Pharmacy and Pharmaceutical Sciences, 8, 138-42.
Kalam, M. A., Khan, A. A., Khan, S., Almalik, A., & Alshamsan, A. (2016). Optimizing indomethacin-loaded chitosan nanoparticle size, encapsulation, and release using Box–Behnken experimental design. International Journal of Biological Macromolecules, 87, 329-340. https://doi.org/10.1016/j.ijbiomac.2016.02.033
Kareem, O., Ali, T., Dar, L. A., Mir, S. A., Rashid, R., Nazli, N., ... & Bader, G. N. (2022). Positive health benefits of saponins from edible legumes: Phytochemistry and pharmacology. Edible Plants in Health and Diseases: Volume II: Phytochemical and Pharmacological Properties, 279-298. https://doi.org/10.1007/978-981-16-4959-2_8
Kolekar, C., Patil, S. P., & Kumbhar, S. T. (2019). Synthesis and human RBC membrane stabilization activity of substituted 3-benzoyl flavone. Asian Journal of Pharmacy and Pharmacology, 5(4), 673-677. https://doi.org/10.31024/ajpp.2019.5.4.4
Liang, J., Yan, H., Puligundla, P., Gao, X., Zhou, Y., & Wan, X. (2017). Applications of chitosan nanoparticles to enhance absorption and bioavailability of tea polyphenols: A review. Food Hydrocolloids, 69, 286–292. https://doi.org/10.1016/j.foodhyd.2017.01.041
Mehmood, T., Liland, K. H., Snipen, L., & Sæbø, S. (2012). A review of variable selection methods in partial least squares regression. Chemometrics and Intelligent Laboratory Systems, 118, 62-69. https://doi.org/10.1016/j.chemolab.2012.07.010
Mudalip, S. A., Khatiman, M. N., Hashim, N. A., Man, R. C., & Arshad, Z. I. M. (2021). A short review on encapsulation of bioactive compounds using different drying techniques. Materials Today: Proceedings, 42(1), 288-296. https://doi.org/10.1016/j.matpr.2021.01.543
Munteanu, I. G., & Apetrei, C. (2021). Analytical methods used in determining antioxidant activity: A review. International Journal of Molecular Sciences, 22(7), Article 3380. https://doi.org/10.3390/ijms22073380
Nguyen, M. H., Ha, D. L., Do, B. M., Chau, N. T. N., Tran, T. H., Le, N. T. H., & Le, M. T. (2023). RP-HPLC-based flavonoid profiling accompanied with multivariate analysis: an efficient approach for quality assessment of Houttuynia cordata Thunb leaves and their commercial products. Molecules, 28(17), Article 6378. https://doi.org/10.3390/molecules28176378
Patthamasopasakul, R., Songsak, T., Kunaratnpruk, S., & Sucontphunt, A. (2024). Comparative study: Extraction conditions and antioxidant and antibacterial activities of Gracilaria fisheri. Journal of Current Science and Technology, 14(3), Article 52. https://doi.org/10.59796/jcst.V14N3.2024.52
Permanadewi, I., Kumoro, A. C., Wardhani, D. H., & Aryanti, N. (2022). Effect of viscosity on iron encapsulation using alginate as a carrying agent in a controlled spray drying process. Food Research, 6(5), 56-67. https://doi.org/10.26656/fr.2017.6(5).613
Rachmawati, M., Murdianto, W., Suprapto, H., Apriadi, R., Ramadhani, N. E., Apriliato, D. K., ... & Saragih, B. (2024). Effect of drying methods and harvest age on the quality of Sliced Tiwai Onion (Eleutherine americana Merr) as A Potential Functional Food. Trends in Sciences, 21(12), 8401-8401. https://doi.org/10.48048/tis.2024.8401
Rahmadi, A., Nurjannah, S., Andriyani, Y., Banin, M. M., Rohmah, M., Amaliah, N., ... & Emmawati, A. (2022). Proximate analysis of the high phytochemical activity of encapsulated Mandai cempedak (Artocarpus champeden) vinegar prepared with maltodextrin and chitosan as wall materials. F1000Research, 11, Article 865. https://doi.org/10.12688/f1000research.109612.1
Rahman, Z., Zidan, A. S., Habib, M. J., & Khan, M. A. (2010). Understanding the quality of protein loaded PLGA nanoparticles variability by Plackett–Burman design. International Journal of Pharmaceutics, 389(1-2), 186-194. https://doi.org/10.1016/j.ijpharm.2009.12.040
Raj, R. A., & Murugesan, S. (2022). Optimization of dielectric properties of pongamia pinnata methyl ester for power transformers using response surface methodology. IEEE Transactions on Dielectrics and Electrical Insulation, 29(5), 1931-1939. https://doi.org/10.1109/TDEI.2022.3190257
Ren, X., Hou, T., Liang, Q., Zhang, X., Hu, D., Xu, B., ... & Ma, H. (2019). Effects of frequency ultrasound on the properties of zein-chitosan complex coacervation for resveratrol encapsulation. Food Chemistry, 279, 223-230. https://doi.org/10.1016/j.foodchem.2018.11.025
Russo, C., Stanzione, F., Tregrossi, A., & Ciajolo, A. (2014). Infrared spectroscopy of some carbon-based materials relevant in combustion: Qualitative and quantitative analysis of hydrogen. Carbon, 74, 127-138. https://doi.org/10.1016/j.carbon.2014.03.014
Santos-Sánchez, N. F., Salas-Coronado, R., Villanueva-Cañongo, C., & Hernández-Carlos, B. (2019). Antioxidant Compounds and Their Antioxidant Mechanism. IntechOpen. https://doi.org/10.5772/intechopen.85270
Shahidi, F., & Yeo, J. (2016). Insoluble-bound phenolics in food. Molecules, 21(9), Article 1216. https://doi.org/10.3390/molecules21091216
Siddique, I. (2024). Exploring functional groups and molecular structures: A comprehensive analysis using FTIR spectroscopy. Chemistry Research Journal, 9(2), 70-76. https://doi.org/10.2139/ssrn.4886526
Soltanzadeh, M., Peighambardoust, S. H., Ghanbarzadeh, B., Mohammadi, M., & Lorenzo, J. M. (2021). Chitosan nanoparticles as a promising nanomaterial for encapsulation of pomegranate (Punica granatum L.) peel extract as a natural source of antioxidants. Nanomaterials, 11(6), Article 1439. https://doi.org/10.3390/nano11061439
Sulaiman, S. F., Yusoff, N. A. M., Eldeen, I. M., Seow, E. M., Sajak, A. A. B., & Ooi, K. L. (2011). Correlation between total phenolic and mineral contents with antioxidant activity of eight Malaysian bananas (Musa sp.). Journal of Food Composition and Analysis, 24(1), 1-10. https://doi.org/10.1016/j.jfca.2010.04.005
Sultana, S., Hossain, M. L., Sostaric, T., Lim, L. Y., Foster, K. J., & Locher, C. (2024). Investigating flavonoids by HPTLC analysis using aluminium chloride as derivatization reagent. Molecules, 29(21), Article 5161.
Suwanpitak, K., Sangnim, T., Sriamornsak, P., Puri, V., Sharma, A., & Huanbutta, K. (2025). Development and validation of a reliable reverse-phase high-performance liquid chromatography method for quantifying triterpenes in Centella asiatica: A step towards quality control of herbal products. Science, Engineering and Health Studies, 19, Article 25050008. https://doi.org/10.69598/sehs.19.25050008
Tiwari, R., & Shukla, A. K. (2020). Plant metabolites and their role in health benefits: A brief review. Advance Pharmaceutical Journal, 5(2), 47-53. https://doi.org/10.31024/apj.2020.5.2.2
Turasan, H., Sahin, S., & Sumnu, G. (2015). Encapsulation of rosemary essential oil. LWT-Food Science and Technology, 64(1), 112-119. https://doi.org/10.1016/j.lwt.2015.05.036
Valková, V., Ďúranová, H., Falcimaigne-Cordin, A., Rossi, C., Nadaud, F., Nesterenko, A., ... & Kačániová, M. (2022). Impact of freeze-and spray-drying microencapsulation techniques on β-glucan powder biological activity: A comparative study. Foods, 11(15), Article 2267. https://doi.org/10.3390/foods11152267
Woranuch, S., & Yoksan, R. (2013). Eugenol-loaded chitosan nanoparticles: I. Thermal stability improvement of eugenol through encapsulation. Carbohydrate Polymers, 96(2), 578-585. https://doi.org/10.1016/j.carbpol.2012.08.117
Wunnakup, T., Songsak, T., Vipunngeun, N., & Sueree, L. (2024). Quantitative thin-layer chromatography analysis, antioxidant, and anti-inflammatory activities of polyherbal formulation (Ammarit-Osot) extracts. Journal of Current Science and Technology, 14(3), Article 65. https://doi.org/10.59796/jcst.V14N3.2024.65
Yang, X. J., Glaser, R., Li, A., & Zhong, J. X. (2016a). On the aliphatic versus aromatic content of the carriers of the ‘unidentified’infrared emission features. Monthly Notices of the Royal Astronomical Society, 462(2), 1551-1562. https://doi.org/10.1093/mnras/stw1740
Yang, X., Li, A., Glaser, R., & Zhong, J. (2016b). The C–H stretching features at 3.2–3.5 μm of polycyclic aromatic hydrocarbons with aliphatic sidegroups. The Astrophysical Journal, 825(1), 22. https://doi.org/10.3847/0004-637X/825/1/22
Ydjedd, S., Bouriche, S., López-Nicolás, R., Sánchez-Moya, T., Frontela-Saseta, C., Ros-Berruezo, G., ... & Kati, D. E. (2017). Effect of in vitro gastrointestinal digestion on encapsulated and nonencapsulated phenolic compounds of carob (Ceratonia siliqua L.) pulp extracts and their antioxidant capacity. Journal of Agricultural and Food Chemistry, 65(4), 827-835. https://doi.org/10.1021/acs.jafc.6b05103
Yoksan, R., Jirawutthiwongchai, J., & Arpo, K. (2010). Encapsulation of ascorbyl palmitate in chitosan nanoparticles by oil-in-water emulsion and ionic gelation processes. Colloids and Surfaces B: Biointerfaces, 76(1), 292-297. https://doi.org/10.1016/j.colsurfb.2009.11.007
Yolmeh, M., & Jafari, S. M. (2017). Applications of response surface methodology in the food industry processes. Food and Bioprocess Technology, 10(3), 413-433. https://doi.org/10.1007/s11947-016-1855-2
Zou, Y., Lu, Y., & Wei, D. (2004). Antioxidant activity of a flavonoid-rich extract of Hypericum perforatum L. in vitro. Journal of Agricultural and Food Chemistry, 52(16), 5032-5039. https://doi.org/10.1021/jf049571r
Downloads
Published
How to Cite
License
Copyright (c) 2026 Journal of Current Science and Technology

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.